Unveiling The Secrets Of An Element Rooted In A Continent
An element named after a continent is Europium. Europium is a chemical element with the symbol Eu and atomic number 63. It is a member of the lanthanide series and is named after the continent of Europe.
Europium is a relatively soft, silvery-white metal that is malleable and ductile. It is the most reactive of the lanthanides and readily oxidizes in air. Europium is found in small amounts in some minerals, such as monazite and bastnsite. It is used in various applications, including phosphors, lasers, and nuclear reactors.
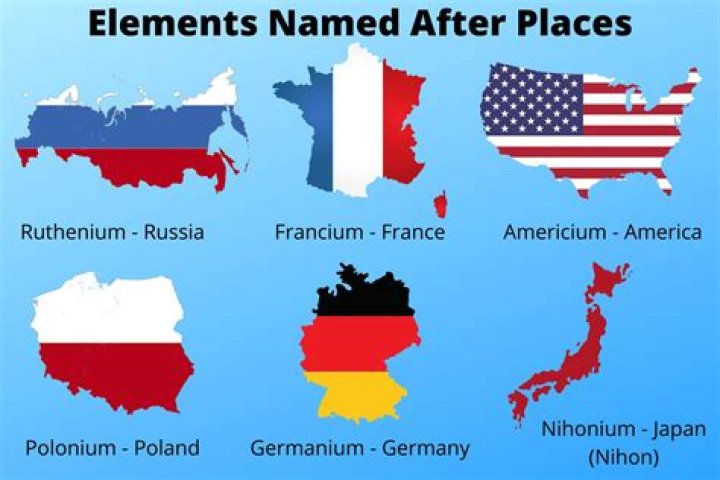
In addition to Europium, there are other two elements named after continents. They are Americium and Francium. Americium is named after the Americas and Francium is named after the France.
One Element Named After a Continent
There are three elements named after continents: Europium, Americium, and Francium. Europium is named after the continent of Europe, Americium is named after the Americas, and Francium is named after France.
- Europium: silvery-white metal
- Americium: radioactive element
- Francium: alkali metal
- All three elements are relatively rare
- Europium is used in phosphors and lasers
- Americium is used in smoke detectors
- Francium is used in medical research
- All three elements are named after places
- Europium is the most common of the three elements
The naming of elements after continents is a relatively recent practice. Europium was the first element to be named after a continent, in 1896. Americium and Francium were named in 1944 and 1939, respectively.
The naming of elements after continents is a way to honor the places where these elements were discovered or first studied. It is also a way to recognize the contributions of the scientists who discovered these elements.
Europium
Europium is a chemical element with the symbol Eu and atomic number 63. It is a member of the lanthanide series and is named after the continent of Europe. Europium is a relatively soft, silvery-white metal that is malleable and ductile. It is the most reactive of the lanthanides and readily oxidizes in air. Europium is found in small amounts in some minerals, such as monazite and bastnasite. It is used in various applications, including phosphors, lasers, and nuclear reactors.
- Facet 1: Properties of Europium
Europium is a silvery-white metal that is soft, malleable, and ductile. It is the most reactive of the lanthanides and readily oxidizes in air. Europium has a melting point of 822 C and a boiling point of 1597 C.
- Facet 2: Applications of Europium
Europium is used in a variety of applications, including phosphors, lasers, and nuclear reactors. Europium-activated phosphors are used in fluorescent lamps and television screens. Europium lasers are used in medical applications, such as laser surgery and laser cutting. Europium is also used in nuclear reactors as a neutron absorber.
- Facet 3: Production of Europium
Europium is produced by reducing europium oxide with calcium or lanthanum. The resulting europium metal is then purified by electrolysis.
- Facet 4: Europium and other elements named after continents
Europium is one of three elements named after continents. The other two elements are Americium and Francium. Americium is named after the Americas and Francium is named after France.
Europium is a versatile element with a wide range of applications. It is an important component of many modern technologies, and its unique properties make it a valuable material for research and development.
Americium
Americium is a radioactive element with the symbol Am and atomic number 95. It is a member of the actinide series and is named after the Americas. Americium is a relatively soft, silvery-white metal that is malleable and ductile. It is the most reactive of the actinides and readily oxidizes in air. Americium is found in small amounts in uranium ores and is produced as a byproduct of nuclear reactions. It is used in various applications, including smoke detectors, neutron sources, and medical imaging.
Americium is one of three elements named after continents. The other two elements are Europium and Francium. Americium is named after the Americas because it was first discovered in the United States in 1944. Americium is a versatile element with a wide range of applications. It is an important component of many modern technologies, and its unique properties make it a valuable material for research and development.
One of the most important applications of americium is in smoke detectors. Americium-241 is used as the radioactive source in ionization smoke detectors. When smoke particles enter the ionization chamber, they interact with the americium-241 and cause the release of electrons. These electrons then create an electrical current that triggers the alarm.
Americium-241 is also used in neutron sources. Neutron sources are used in a variety of applications, including oil well logging, mineral exploration, and medical imaging. Americium-241 produces neutrons when it undergoes alpha decay. These neutrons can then be used to bombard other materials, which can cause the release of gamma rays. Gamma rays are used in a variety of applications, including medical imaging and cancer treatment.Francium
Francium is an alkali metal with the symbol Fr and atomic number 87. It is the heaviest alkali metal and the second-rarest naturally occurring element, after astatine. Francium is named after France, where it was first discovered in 1939.
- Facet 1: Properties of Francium
Francium is a soft, silvery-white metal that is highly reactive. It is the most electropositive element and the most reactive of all the alkali metals. Francium reacts vigorously with water and oxygen, and it tarnishes quickly in air. Francium has a melting point of 27 C and a boiling point of 677 C.
- Facet 2: Applications of Francium
Francium has very few practical applications due to its rarity and high radioactivity. However, it is used in some scientific research, such as in the study of atomic physics and nuclear chemistry.
- Facet 3: Production of Francium
Francium is produced by the decay of actinium-227. Actinium-227 is a radioactive element that is found in uranium ores. Francium can be isolated from actinium-227 by chemical means.
- Facet 4: Francium and other elements named after continents
Francium is one of three elements named after continents. The other two elements are Europium and Americium. Europium is named after the continent of Europe, and Americium is named after the Americas.
Francium is a fascinating element with unique properties. It is the heaviest alkali metal and the most reactive of all the elements. Francium has very few practical applications, but it is used in some scientific research. Francium is also one of three elements named after continents, which highlights its importance in the history of science.
All three elements are relatively rare
The three elements named after continents, Europium, Americium, and Francium, are all relatively rare. This is because they are all produced by nuclear reactions, either naturally occurring or man-made. Europium is the most common of the three, with an abundance of about 2 parts per million in the Earth's crust. Americium is less common, with an abundance of about 0.05 parts per million. Francium is the rarest of the three, with an abundance of only about 10^-18 parts per million.
- Facet 1: The rarity of europium
Europium is the most common of the three elements named after continents, but it is still relatively rare. It is found in small amounts in some minerals, such as monazite and bastnasite. Europium is used in a variety of applications, including phosphors, lasers, and nuclear reactors.
- Facet 2: The rarity of americium
Americium is less common than europium. It is found in small amounts in uranium ores and is produced as a byproduct of nuclear reactions. Americium is used in a variety of applications, including smoke detectors, neutron sources, and medical imaging.
- Facet 3: The rarity of francium
Francium is the rarest of the three elements named after continents. It is only found in trace amounts in uranium ores. Francium has very few practical applications due to its rarity and high radioactivity.
The rarity of these elements makes them valuable for scientific research. They are used in a variety of applications, including nuclear energy, medicine, and space exploration. The study of these elements has also led to a greater understanding of the universe and our place in it.
Europium is used in phosphors and lasers
Europium is one of three elements named after continents. It is a versatile element with a wide range of applications, including in phosphors and lasers. Phosphors are materials that emit light when they are exposed to radiation. Europium-activated phosphors are used in fluorescent lamps and television screens. Lasers are devices that emit a concentrated beam of light. Europium lasers are used in medical applications, such as laser surgery and laser cutting.
The use of europium in phosphors and lasers is important because it allows us to create devices that emit light in a specific color or wavelength. This is essential for many applications, such as medical imaging and laser cutting. Europium-activated phosphors are also used in energy-efficient lighting, which can help to reduce our reliance on fossil fuels.
The study of europium and its applications has led to a greater understanding of the universe and our place in it. Europium is a fascinating element with unique properties that make it valuable for a wide range of applications. By continuing to study europium and other elements, we can continue to develop new technologies that will benefit society.
Americium is used in smoke detectors
Americium, an element named after the Americas, plays a crucial role in ensuring our safety through its application in smoke detectors. This remarkable invention safeguards lives by detecting the presence of smoke, a telltale sign of fire, and triggering an alarm to alert occupants.
- Facet 1: Ionization chambers and americium
Smoke detectors utilize ionization chambers, where americium-241, a radioactive isotope, ionizes air molecules. When smoke particles enter the chamber, they disrupt the ionization process, causing a drop in current and triggering the alarm.
- Facet 2: Americium's unique properties
Americium's radioactivity is precisely what makes it suitable for smoke detectors. Its alpha particles have a short range and interact effectively with air molecules, creating ions necessary for the ionization process.
- Facet 3: Safety considerations
Americium's radioactivity raises concerns about safety. However, smoke detectors are designed with multiple layers of containment to prevent americium from escaping. Regular maintenance and proper disposal ensure minimal exposure and environmental impact.
- Facet 4: Americium and other smoke detector types
Americium is primarily used in ionization smoke detectors. Other types, such as photoelectric detectors, use different mechanisms to detect smoke particles, complementing americium-based detectors for comprehensive fire safety.
In conclusion, americium's presence in smoke detectors exemplifies the ingenuity of science in harnessing the unique properties of elements. By leveraging americium's radioactivity, we enhance our safety and preparedness against the dangers of fire.
Francium is used in medical research
Francium, an element named after France and the heaviest alkali metal, holds a unique place in medical research due to its radioactive properties and the insights it offers into various biological processes.
- Facet 1: Francium-223 and targeted alpha therapy
Francium-223, a radioactive isotope of francium, has shown promising results in targeted alpha therapy for treating certain types of cancer. Alpha particles emitted by francium-223 can precisely target and destroy cancer cells while minimizing damage to surrounding healthy tissue.
- Facet 2: Francium as a tracer in PET imaging
Francium-218, another radioactive isotope, serves as a valuable tracer in positron emission tomography (PET) imaging. PET scans using francium-218 allow medical professionals to visualize and assess metabolic processes in the body, aiding in the diagnosis and monitoring of various diseases.
- Facet 3: Francium in bone marrow imaging
Francium-223 has also found applications in bone marrow imaging, where it helps visualize and evaluate bone marrow activity. This technique is particularly useful in diagnosing and monitoring conditions affecting the bone marrow, such as leukemia and other blood disorders.
- Facet 4: Francium's contribution to nuclear medicine
Beyond its direct medical applications, francium's unique properties have contributed to the advancement of nuclear medicine. Its radioactive isotopes serve as calibration sources for PET scanners, ensuring accurate and reliable imaging results.
In conclusion, francium, an element named after a continent, plays a significant role in medical research, particularly in targeted cancer therapy, PET imaging, and bone marrow imaging. Its unique radioactive properties offer valuable insights into biological processes and contribute to the development of innovative diagnostic and therapeutic tools.
All three elements are named after places
The three elements named after continents, Europium, Americium, and Francium, share a unique characteristic: they are all named after places. This connection highlights the historical and geographical context surrounding the discovery and naming of these elements, offering insights into scientific exploration and the recognition of diverse regions.
- Recognition of geographical diversity
The naming of elements after continents acknowledges the global nature of scientific discovery. By associating elements with different parts of the world, it reflects the contributions of scientists from various regions and celebrates the diversity of our planet.
- Commemorating scientific achievements
The act of naming elements after places serves as a tribute to the scientific achievements associated with those locations. For instance, Europium's name honors the discovery of the element in Europe, recognizing the contributions of European scientists to the field of chemistry.
- Inspiration for further exploration
The connection between elements and places can inspire further scientific exploration and research. It encourages scientists to investigate the unique properties and characteristics of elements found in different regions, potentially leading to new discoveries and applications.
- Cultural and historical significance
The naming of elements after continents carries cultural and historical significance. It reflects the influence of geography and place on scientific nomenclature, providing a glimpse into the values and perspectives of the time when these elements were discovered.
In conclusion, the fact that all three elements named after continents are also named after places underscores the global nature of scientific discovery and the recognition of geographical diversity in the field of chemistry. It serves as a reminder of the importance of honoring scientific achievements, inspiring further exploration, and acknowledging the cultural and historical context surrounding the naming of elements.
Europium is the most common of the three elements
Among the three elements named after continents, Europium stands out as the most common. This distinction holds significance in understanding the prevalence and accessibility of these elements, as well as their potential applications.
The abundance of Europium compared to Americium and Francium influences its availability for scientific research and practical uses. Its relatively higher concentration in the Earth's crust makes it more feasible to extract and utilize Europium for various purposes. This abundance also contributes to the element's affordability, making it a more viable option for large-scale applications.
The prevalence of Europium among the three continental elements has practical implications in diverse fields. For instance, Europium's use in phosphors and lasers benefits industries such as lighting, display technologies, and medical devices. Its role in nuclear reactors highlights its importance in energy production and research. The accessibility of Europium allows for the exploration of its unique properties and the development of innovative applications across scientific disciplines.
Understanding the relative abundance of Europium deepens our knowledge of the distribution and availability of elements on Earth. It guides scientists and researchers in selecting appropriate materials for specific applications, considering factors such as cost, availability, and environmental impact. Moreover, it encourages the exploration of alternative sources and extraction methods to ensure the sustainable use of these valuable elements.
FAQs on "One Element Named After a Continent"
This section addresses frequently asked questions regarding elements named after continents, providing concise and informative answers to enhance understanding.
Question 1: Which element is named after a continent?
Answer: Europium is the element named after the continent of Europe.
Question 2: Why are elements named after continents?
Answer: Elements are named after continents to honor the locations where they were discovered or to recognize the contributions of scientists from those regions.
Question 3: What is the significance of naming elements after continents?
Answer: It acknowledges the global nature of scientific discovery and celebrates the diversity of our planet, inspiring further exploration and research.
Question 4: How many elements are named after continents?
Answer: Three elements are named after continents: Europium, Americium, and Francium.
Question 5: Which continent has the most elements named after it?
Answer: Europe is the only continent with an element named after it (Europium).
Question 6: What are some practical applications of elements named after continents?
Answer: Europium is used in phosphors and lasers, Americium in smoke detectors and neutron sources, and Francium in medical research and nuclear medicine.
In summary, the naming of elements after continents highlights the interconnectedness of science and geography, recognizes scientific achievements, and encourages further exploration and innovation.
Transition to the next article section:
Tips on Understanding "One Element Named After a Continent"
Exploring the concept of elements named after continents requires a multifaceted approach. Here are a few tips to enhance your understanding:
Tip 1: Explore the Historical Context
Delve into the historical background of the discoveries and namings of these elements. Understanding the motivations and scientific advancements of the time can provide insights into the significance of these elements.
Tip 2: Examine Properties and Applications
Investigate the unique properties and practical applications of each element named after a continent. Learn about their uses in fields such as technology, medicine, and energy production.
Tip 3: Consider Geographical Significance
Recognize the geographical significance behind the naming of these elements. The connection to specific continents highlights the global nature of scientific discovery and the contributions of diverse regions.
Tip 4: Trace the Evolution of Nomenclature
Follow the evolution of the nomenclature of these elements. Understanding how their names have changed over time can provide insights into the development of scientific understanding.
Tip 5: Discuss with Experts
Engage with scientists, historians, or educators specializing in this topic. Their knowledge and perspectives can enrich your comprehension and provide valuable insights.
Summary: By following these tips, you can gain a deeper understanding of the elements named after continents, their historical significance, practical applications, and the fascinating interplay between science and geography.
Transition to the article's conclusion:
Conclusion
Our exploration of "one element named after a continent" has highlighted the fascinating interplay between science and geography. The naming of elements after continents acknowledges the global nature of scientific discovery and celebrates the contributions of diverse regions to our understanding of the world.
Recognizing the importance of understanding the elements named after continents goes beyond mere knowledge acquisition. It invites us to appreciate the rich history of scientific exploration, the practical applications of these elements, and their significance in fields ranging from technology to medicine. Embracing this knowledge empowers us to make informed decisions, foster innovation, and contribute to the advancement of scientific endeavors.
Remi Wolf: Unveiling The Secrets Of Her Age And Success
Unveiling The Private World Of Ricky Williams' Son: Exclusive Insights
Unlocking Education's Potential: Discoveries With Bernadette San Pedro Bayot